First Class Info About How To Find Out The Atomic Mass Of An Element

The average atomic mass of elements can be calculated by the abundance of each.
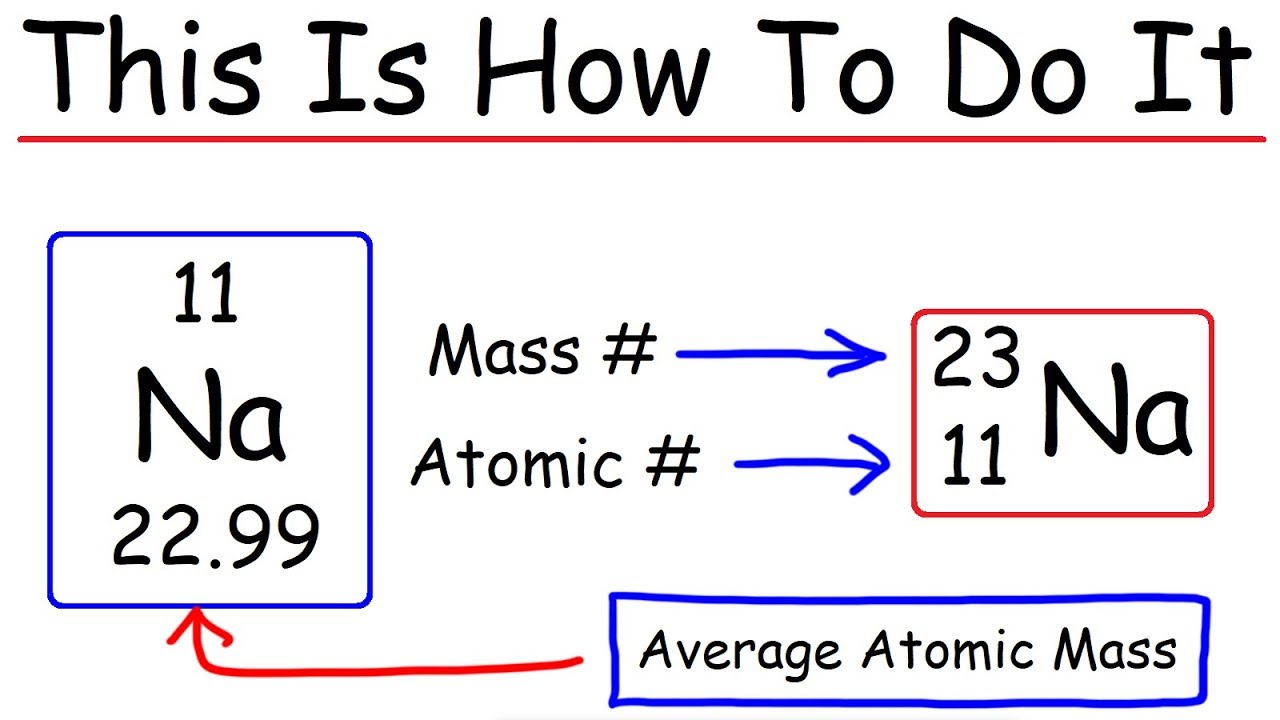
How to find out the atomic mass of an element. The relative atomic mass of an element is the total mass of the element's naturally occurring isotopes. Get the full course at: To obtain the atomic mass of a particular atom, you simply add the number of protons and the number of neutrons present in the atom.
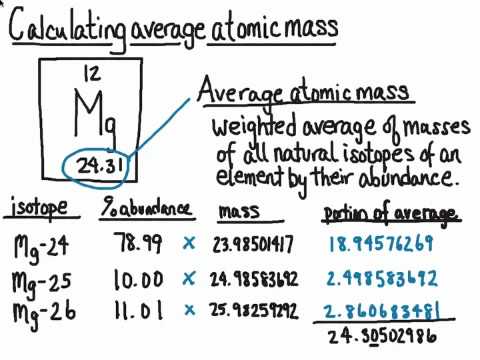
Atomic mass = number of protons + number of neutrons. How to calculate average atomic mass. For each isotope, multiply its mass by the.
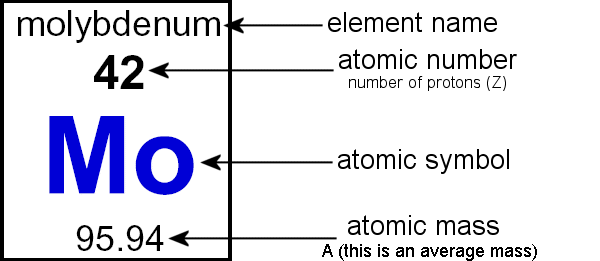
The formula that is generally used to calculate the atomic mass for an element is as follows. A = n + z. The sum of the masses of protons, neutrons, and electrons in an atom is the atomic mass.
Atomic mass calculator results (detailed calculations. Identify the percentage of each isotope in the composition of the element and its mass. This is the mass of the atom divided by its charge number.
In contrast with protons or neutrons, the electrons have much less mass, so the mass of. Method 1, sum total of protons and neutrons of a single atom, these steps. To find the atomic mass of a given element or an isotope of an element, there are 3 steps involved:
How to calculate the atomic mass of an atom, a specific isotope’s atomic mass corresponds to its total mass expressed in dalton (u), also called unified atomic mass units. For example, consider an oxygen atom having 8. Here, a is the mass.
Atomic mass number is the number of protons and neutrons in the nucleus. There are three main ways to calculate atomic mass. Standard atomic mass is determined by the average weight of all the isotopes of a given sample element.
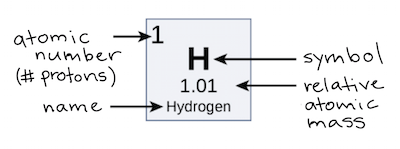
Atomic number is the number of protons in the nucleus. For neutral atoms, the charge.


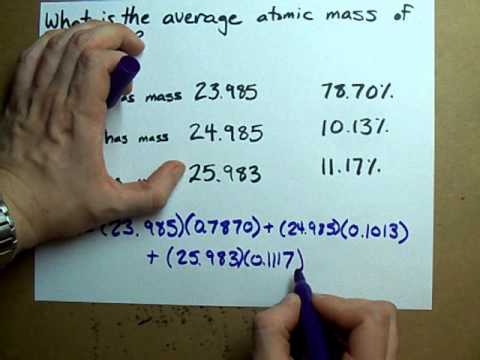


/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)



/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)


/atomic-mass--58dc0d885f9b58468332c41b.jpg)